Wei Weifeng AM, Central South University: Salt-assisted recovery of sodium metal anode for high-rate sodium batteries
Background
Rechargeable sodium metal batteries are considered to be one of the most promising electrochemical energy storage systems with high energy density and high cost performance. However, the inactive sodium (Na) formed during storage and assembly processes has seriously hindered its practical application. This chemical instability stems from the high activity of Na, which chemically reacts with oxygen and moisture during electrode processing or electrochemically reacts with the electrolyte during battery operation, easily leading to excessive accumulation of inactive Na species on the surface, resulting in battery performance degradation or even failure.
Introduction
In this work, we designed a simple and feasible method to simultaneously recycle the deactivated Na anode and enhance the ionic conductivity of the SEI by modifying the Na metal surface with 3-glycolyloxypropyltrimethoxysilane (GPTMS). By coating GPTMS on Na metal (GPTMS@Na), the inert layer can be eliminated, thereby achieving electrochemical activity recovery on pristine and air-exposed Na anode. The metallophilic GPTMS molecules are grafted onto Na metal via Si─O─Na bonds and form a self-assembled bilayer with external hydrophobic groups via polycondensation of siloxane and ring-opening polymerization of epoxide. As a result, the electrochemical activity of the Na metal anode can be fully restored, and the robust GPTMS-derived SEI layer ensures high capacity and long-term cycling at an ultrahigh rate of 30 C (93.1 mAh g-1, 94.8% after 3000 cycles). Through this salt-assisted modification strategy, the exposed Na metal can not only achieve electrochemical recovery but also withstand humid air attack and high current density cycling. This article was published in Advanced Materials with the title “ Salt-Assisted Recovery of Sodium Metal Anodes for High-Rate Capability Sodium Batteries ”. The first author is Lin Jialin and the corresponding author is Wei Weifeng from Central South University.
Main content
Figure 1. GPTMS recovery treatment. a) Schematic diagram of the GPTMS surface treatment process. b) Optical images of two Na foils exposed to humid air and treated with GPTMS. c) FTIR spectra and d, e) SEM secondary electron (SE) images of different Na samples.
Figure 2. Characterization of GPTMS@Na. a) XRD patterns of pristine Na and GPTMS@Na. b) FTIR spectra of pure GPTMS, Na, and GPTMS@Na. c, d) XPS spectra of pristine Na and e, f) GPTMS@Na. g) TOF-SIMS depth profiles of pristine Na and GPTMS@Na and visualization of their spatial distribution.
Figure 3. Electrochemical performance. a) Voltage profile of Na||Na battery and b) long-term cycling performance of NVP||Na battery with different electrodes. c) Voltage profiles of pristine Na||Na and GPTMS@Na|| GPTMS@Na batteries measured at current densities of 3 mA cm -2 and d) 0.125 to 4 mA cm -2 . e) Rate capacity and cycling performance of NVP||Na battery with different anodes. g) Rate capacity and h) cycling performance of NFM||Na battery with different anodes.
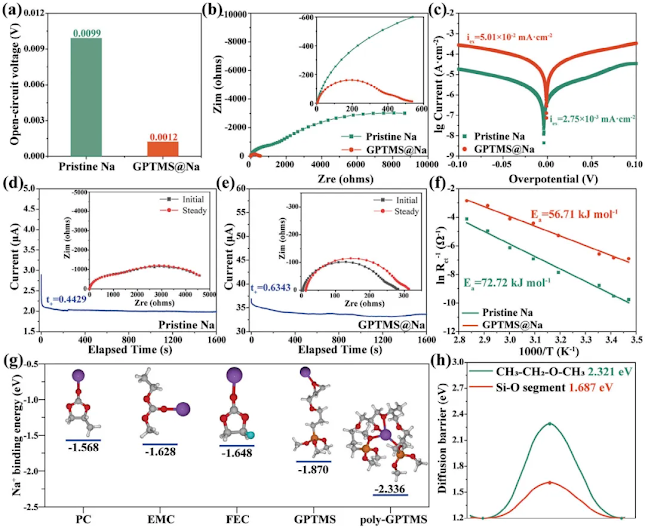
Figure 4. Interface dynamics . a) OCV of Na||Na symmetric cell before and after GPTMS treatment. b) Nyquist plot and c) Tafel plot of different electrodes. Current evolution over time when polarizing Na||Na cell using d) pristine Na and e) GPTMS@Na at 10 mV. Inset corresponds to the impedance spectra before and after polarization. f) Arrhenius curves and activation energy comparison of different electrodes. g) Optimized structures of the studied molecules and their adsorption structures on Na + , where white, gray, red, cyan, purple, and orange represent H, C, O, F, Na, and Si atoms, respectively. h) Diffusion barriers of each segment.
Figure 5. Chemical evolution of SEI. In situ FTIR spectra of a) pristine Na and b) GPTMS@Na during the first two cycles. c, d) Element ratio changes, e, f) C1s spectra of pristine Na and GPTMS@Na anodes at different sputtering depths after 100 cycles, g, h) F 1s spectra.
in conclusion
In summary, this work demonstrates an effective salt-assisted recycling strategy to construct a hydrophobic bilayer with excellent ionic conductivity while activating the deactivated Na metal through the spontaneous hydrolysis and polycondensation of GPTMS. The self-assembled protective SEI is beneficial for regulating the deposition behavior of Na and promoting uniform Na deposition/stripping, thereby improving the cycling stability of the Na metal anode at an ultrahigh rate of 30 C. Therefore, the GPTMS treatment ensures the electrochemical kinetics, cycling stability, and stability of the Na metal anode under humid air. This work provides a simple and effective route for the recycling and development of practical next-generation small and medium-sized metal batteries and other alkali metal batteries.
References
Salt-Assisted Recovery of Sodium Metal Anodes for High-Rate Capability Sodium Batteries. Adv. Mater. 2024, 2409976
Full text link:
DOI: 10.1002/adma.202409976
Canrd Brief Introduce
Canrd use high battery R&D technology(core members are from CATL) and strong Chinese supply chain to help many foreign companies with fast R&D. We provide lab materials,electrodes, custom dry cells, material evaluation, perfomance and test, coin/pouch/cylindrical cell equipment line, and other R&D services.
Email:janice@canrd.com
Phone/Wechat/WhatsApp/Skype:+86 18928276992
Website : www.canrud.com







No comments:
Post a Comment